Peste des petits ruminants in Europe: the 2024–2025 alert and the evolving landscape of surveillance
Introduction
Peste des petits ruminants (PPR) is a highly contagious viral disease primarily affecting sheep and goats, although other domestic and wild ruminants may also be involved. It is caused by a virus belonging to the genus Morbillivirus (family Paramyxoviridae), phylogenetically related to the rinderpest virus, with 4 genetic lineages (I–IV) being identified. Clinical signs of disease include fever, depression, oral lesions, oculonasal discharge, respiratory symptoms, and diarrhoea, with high mortality rates generally ranging from 20% to 90%, and approaching 100% in susceptible populations1.
With estimated losses ranging from €1.3 to €2 billion per year (Food and Agricolture organization of the United Nations, FAO)3, the eradication of PPR by 2030 represents a shared global objective. This goal is part of the joint FAO-World Animal health Organisation (WOAH) initiative for global eradication, based on integration of vaccination, diagnostics, and strengthening of veterinary services.
PPR is mainly transmitted through direct contact between infected animals via secretions and excretions, particularly respiratory secretions; indirect transmission via fomites is possible but less relevant. The incubation period is typically 5–6 days, during which the virus may already be shed.
From an epidemiological perspective, animal movements play a key role in the spread of PPR, particularly in the context of informal and cross-border trade, which may enable the introduction of the virus into new areas and susceptible populations. Under such conditions, the disease can spread rapidly, leading to outbreaks characterized by high morbidity and mortality.
Diagnosis is based on clinical and epidemiological evaluation, followed by laboratory confirmation. This can be performed using RT-PCR, considered the gold standard for diagnostic confirmation, serological tests (ELISA), and virus isolation and genetic sequencing. Suitable samples include nasal, ocular, and rectal swabs, whole blood in EDTA, serum, organs (lymph nodes, lung, spleen), and whole carcasses2.
Within the European regulatory framework, PPR is classified as a Category A disease, meaning it should not be present within the European Union and that it requires immediate eradication measures upon confirmation. These include compulsory culling of all susceptible animals in the infected establishment, followed by safe disposal of carcasses as Category 1 material under Regulation (EC) No. 1069/20094. In parallel, restriction zones are established around the outbreak: a protection zone (minimum radius 3 km) and a surveillance zone (minimum radius 10 km), within which strict movement restrictions and intensive screening through clinical examinations and laboratory tests are applied.
Operational provisions for managing suspicion and confirmation, establishing restriction zones, and applying measures are defined within the Animal Health Law framework. In particular, Regulation (EU) 2016/4295 provides the general legal framework, while Delegated Regulation (EU) 2020/6876 defines prevention and control measures for Category A diseases, including PPR.
Although not a zoonosis, PPR has significant sanitary and economic impact due to production losses, direct and indirect costs, and trade restrictions implemented to control its spread.
Traditionally considered a major threat in Africa, the Middle East, and Asia - with increasing predominance of lineage IV7 - the emergence of outbreaks in Bulgaria in 2018 and subsequent incursions into Greece and Romania in 2024 have highlighted the vulnerability of the European Union and the tangible risk of further spread in other countries in the region. This situation requires urgent revision of prevention, control, and eradication strategies.
This article examines the sequence of incursions that have progressively reshaped the epidemiological situation, the new knowledge guiding the health response, and the evolution of surveillance systems aimed at protecting countries that remain free from infection, such as Italy.
Epidemiological situation in Europe: developments since 2024
PPR was first described in 1942 in Côte d’Ivoire. In Europe, the first confirmed case was reported in 2000 in Eastern Thrace (the European part of Turkey)2.
The perception of Europe as largely free from PPR has been significantly challenged during the 2024–2025 period. While earlier events, such as the 2018 outbreak in Bulgaria, could be considered isolated incursions, recent developments indicate a changing epidemiological landscape: PPR has established an active presence in south-eastern Europe, with a risk of further consolidation and geographic expansion.
In July 2024, the disease was reported for the first time in Greece and shortly after in Romania. These events were followed throughout 2024-2025 by further outbreaks in Bulgaria, Hungary, Albania, and Croatia, countries within or bordering the EU that were previously free from the disease.
2024–2025: sequence of incursions in south-eastern Europe8,9
Greece (2024)10,11: in July, the first outbreak was confirmed in Thessaly, in north-central Greece, in the regional units of Trikala and Larisa. Subsequently, dozens of outbreaks were notified in other regions of the country. Thessaly is home to approximately 1.7 million small ruminants distributed across 8,500 farms, with over 1 million animals concentrated in the regional unit of Larisa alone. This high density, combined with the widespread practice of mixed grazing, facilitated rapid viral spread.
The delay in diagnosing the first outbreak, initially mistaken for bluetongue, allowed the virus to spread before the implementation of sanitary restrictions. Despite the subsequent adoption of control measures, including the establishment of restriction zones, disinfection, movement controls, and field surveillance, transmission continued.
In November 2024, when the emergency was considered resolved, Greece reported new outbreaks in the regional units of Corinthia and Elis, in the southern part of the country. Epidemiological investigations identified several critical factors contributing to the spread of the epidemic, including intense animal trade, biosecurity gaps (particularly in milk collection trucks), and animal movements occurring prior to the implementation of restrictions, when some animals were already in the incubation period.
Romania (2024)12,13: the first PPR outbreaks were notified in July 2024 in the counties of Tulcea and Constanța, in the south-eastern part of the country. In this case, the size of the affected farms was particularly significant: among the infected holdings, one exceeded 50,000 sheep, while two others had 12,000 and 19,000 head respectively. Within a few days, the virus spread for over 500 kilometres and reached Timiș County, in the western extremity of the country. In August 2024, a new outbreak was confirmed in Ialomița County, 80 kilometers southwest of the initial cluster in Tulcea. The European Commission, after revising the restriction zones for the second time, reported a very high risk of further spread of the infection. Following the closure of this first epidemic event, a new outbreak was notified in March 2025, associated with the emergence of outbreaks in the Crișana region, in the northwest.
Bulgaria (2024): at the end of November, Bulgaria officially notified a PPR outbreak in the Pazardzhik region, in the south of the country, although the first clinical signs had been observed several weeks earlier. The infection involved five farms belonging to the same family, with animals raised under extensive conditions and practicing. The farmers appealed to the court against the culling measures, obtaining their suspension; however, a second round of sampling confirmed the animals' positivity. The event triggered a strong negative reaction from public opinion, further complicating the implementation of control measures. Clinical and laboratory investigations carried out on over 8,000 animals in epidemiologically linked areas did not detect further outbreaks. Bulgarian authorities subsequently established 5 km protection and 20 km surveillance zones. Despite the measures adopted, the outbreak remains active, and the risk of further spread of the infection in the region remains high.
Hungary (2025)14,15: in January 2025, a PPR outbreak was confirmed in Zala County, located in the south-western extremity of Hungary, just 3 kilometres from the border with Slovenia. The epidemiological investigation traced the likely source of introduction of the virus to a batch of ewe lambs imported from Romania, a country already affected by numerous outbreaks. Phylogenetic analysis placed the Hungarian strain in lineage IV, closely related to strains circulating in Romania, Greece, and Bulgaria. Following the notification, the prescribed containment measures were promptly implemented. Subsequently, two secondary outbreaks epidemiologically linked to the primary outbreak were recorded: one attributed to direct contact between animals, and the other associated with the purchase of hay from a farm involved in the primary outbreak. All infected farms were subjected to total culling and disinfection; operations were completed in February, allowing the outbreak to be declared closed. As a precautionary measure, Slovenia established its own surveillance zone along the border. This episode highlights the potential role of live animal imports from Balkan countries in introducing the virus into previously unaffected areas.
Albania (2025): in June 2025, the first outbreaks were notified, with infections appearing almost simultaneously in several areas of the country: from the district of Shkodër (northwest), to the mountainous districts of Dibër and Bulqizë (northeast), to the inland areas of Skrapar (south-central), and those of Polenë and Devoll (southeast), on the border with Greece. The outbreaks affected only goats; this has led to the hypothesis of a possible subclinical circulation in sheep or a differential susceptibility between the two species. The epidemiological investigation did not definitively identify the source of introduction; however, it is suspected that the virus entered the country through illegal animal movements or the importation of potentially infected lambs. The coincidence with the Kurban Bayram holiday (June 6), a period characterized by increased trading and movement of small ruminants, may have further facilitated the spread of the infection. The implementation of control measures proved complex due to the widespread practice of mixed grazing, the absence of state compensation for culling, and the resulting low level of farmers’ compliance. The report of the European Union Veterinary Emergency Team (EUVET) mission recommends strengthening serological surveillance in sheep, rapidly launching a vaccination strategy, and sending samples to the European Union reference laboratory (at the "Centre de coopération internationale en recherche agronomique pour le développement," Montpellier, France) for genetic characterization of the circulating strain, in order to clarify the origin of the introduction.
Kosovo (2025): due to the extent of disease spread in Albania, Kosovar veterinary authorities strengthened active surveillance, particularly in border areas. In July 2025, the first and only PPR outbreak was confirmed in a mixed farm in the Prizren region, approximately 15 km from the Albanian border, affecting a population of 178 goats and 13 sheep. The introduction of the virus was most likely attributed to illegal movements of small ruminants from Albania. A 3 km protection zone was established; in addition, EUVET recommended the establishment of a 10 km surveillance zone, based on EU standards, as well as the prioritisation of clinical and serological controls, particularly in animals returning from pastures along the Albanian border. This event highlights the importance of strengthened surveillance and close cross-border cooperation to prevent the introduction of the virus into previously disease-free areas.
Croatia (2025): in December, the first introduction of the virus was confirmed in a sheep farm in the municipality of Prgomet, in Split-Dalmatia County, in the south-central part of the country. The infection was identified during differential diagnostic investigations initiated following the appearance of abortive symptoms. The initial outbreak involved 26 sheep, all of which tested seropositive. Authorities ordered the immediate culling of the animals and the establishment of restriction zones, initially of 3 and 10 km, later extended to 5 and 20 km. Between December 2025 and January 2026, four additional outbreaks were notified, all epidemiologically linked to the first, likely through direct animal contact or mixed grazing. Among these, a case in a mixed sheep and goat farm in Zadar County was also reported. Croatian authorities implemented strict control measures, including culling, cleaning and disinfection, systematic veterinary visits in affected areas, and strengthened biosecurity requirements across the national territory. The source of the infection was not definitively identified. However, critical issues emerged related to illegal animal movements and documentation deficiencies along the border with Bosnia and Herzegovina, which are considered the most likely mechanism of introduction. The geographical location of the outbreaks also indicates the presence of the virus on the eastern coastline of the Adriatic Sea.
Overall, this sequence of events does not represent a series of independent introductions, but rather a signal of sustained transmission between contiguous countries. Even the seemingly more contained outbreaks, such as those in Hungary or Croatia, therefore take on significant epidemiological meaning, indicating that the virus is circulating in the region and continues to find pathways for spread through animal movement networks, both formal and informal.
Genetic analyses indicate that the viral strains responsible for the European PPR outbreaks are genetically related to those circulating in North Africa, while any genetic correlation with strains present in neighbouring Turkey has been ruled out. The precise routes and mechanisms of virus introduction remain under investigation, although animal movement routes, including informal or untracked ones, represent the main risk factor for the spread of infection into susceptible populations.
It should also be considered that numerous outbreaks were identified through epidemiological tracing activities and diagnostic testing carried out in restriction zones. Indeed, the mild clinical course of the disease can make its early detection difficult.
Table 1 summarises the affected countries, the temporal evolution of events, and data on morbidity and mortality associated with outbreaks confirmed since 2024
Table 1 . Sequence of events in the European area including the number of outbreaks, cases, deaths, and suspected sources of infection
Data source: WAHIS, updated as of 09/02/20269
| Event start date | Country affected | Confirmed outbreaks | Cases | Deaths | Event closure date | Suspected source |
| 07/08/2024 | Greece | 86 | 5317 | 606 | 01/01/2025 | Unknown or inconclusive |
| 07/15/2024 | Romania (prima comparsa) | 67 | 226162 | 13761 | 12/02/2024 | Unknown or inconclusive |
| 11/25/2024 | Bulgaria | 1 | 25 | 4 | In progress | Unknown or inconclusive |
| 01/23/2025 | Ungheria | 3 | 433 | 30 | 01/31/2025 | Legal animal movements |
| 03/03/2025 | Romania (recurrence) | 1 | 664 | 12 | 04/11/2025 | Unknown or inconclusive |
| 06/03/2025 | Albania (first occurrence in a zone or compartment) |
10 | 271 | 67 | In progress | Unknown or inconclusive |
| 06/04/2025 | Albania (first occurrence in the country) |
3 | 179 | 86 | In progress | Unknown or inconclusive |
| 07/03/2025 | Kosovo | 1 | 50 | 6 | *Not available | Animal movements |
| 12/06/2025 | Croatia | 5 | 26 | 0 | In progress | Unknown or inconclusive |
*Closure date not available. Country not recognized as an independent State by 5 EU Member States. Data source: PAFF Committee
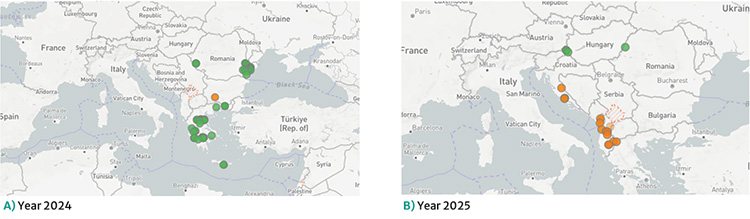 Figure 1. Spatial distribution of outbreaks in south-eastern Europe in 2024 (A) and 2025 (B)
Figure 1. Spatial distribution of outbreaks in south-eastern Europe in 2024 (A) and 2025 (B)
Data source: WAHIS. In green, closed outbreaks; in orange, active outbreaks
Dynamics of spread: the role of molecular epidemiology
Understanding the virus's transmission routes is essential for investigating possible sources of introduction. In this context, molecular epidemiology has established itself as a fundamental tool, capable of transforming isolated individual events into a coherent transmission framework and providing information of immediate operational utility in terms of disease prevention and control.
Phylogenetic studies conducted on the viral strains responsible for the recent incursions indicate a common origin and a close phylogenetic relationship among the viruses isolated in Greece, Romania, and Bulgaria. In particular, the European strains belong to Lineage IV and show a strong genetic correlation with strains circulating in North Africa (Box 1)17.
| Why are genomes useful even in the management of the disease at the territorial level? |
| Genomes that are very similar across distinct outbreaks suggest an epidemiological link or a common source; by contrast, marked differences indicate possible multiple introductions. In operational terms, such information helps guide the prioritisation between actions focused on local containment and actions aimed at strengthening controls on entry points, and animal movements. |
Box 1. Interpretation of genomics
With an estimated probability between 97 and 99%, Romania is the first country affected by the infection16. Genomic analysis of the outbreaks reveals the presence of multiple independent introductions of the virus into Europe, as indicated by the identification of several genetic clusters derived from a common ancestral strain. Furthermore, some of these clusters include sequences from both Romania and Greece, suggesting the existence of subsequent regional transmission chains between different countries, likely facilitated by the movement and trade of infected animals. In interpreting the genomic analysis, several aspects must be considered, including the lack of geographic uniformity in the availability of sequences from outbreaks, the presence of partial data from some epidemiological investigations, and the fact that in some cases samples were obtained several weeks after the onset of the outbreak16.
By revealing near-identity between strains from outbreaks in different countries, genomic analysis represents a tangible support tool for health authorities, allowing them to focus epidemiological investigations on direct contacts and animal movements between areas of specific interest, avoiding the dispersion of resources on generalized control measures. Conversely, genetically distinct strains would indicate separate introduction pathways, shifting attention toward points of entry and international trade.
Prioritising targeted and proportionate interventions thus makes it possible to maximise response effectiveness and optimise the use of resources.
From prevention to control: the European framework against PPR
In light of the real risk of disease introduction, actions aimed at reducing incursion opportunities include strengthening border controls, prudent management of animal introductions, separation of new arrivals, and the adoption of essential biosecurity procedures to limit indirect contact with potentially contaminated vehicles, people, and equipment2.
At the same time, an effective PPR surveillance framework is based on the integration of passive and active surveillance. Passive surveillance relies on the timely detection of clinical signs, while active surveillance is conducted in contexts identified as at risk, such as high-movement areas, markets, and territories adjacent to areas affected by outbreaks.
Regardless of the type of surveillance conducted, in the presence of a suspect case, Real-Time RT-PCR represents the gold standard for diagnostic confirmation, capable of providing a result within a few hours. For confirmed cases, genomic characterization must follow, feeding the molecular epidemiology intelligence system. The synergy between a sensitive surveillance system and a rapid, accurate diagnostic network constitutes the infrastructure for effective emergency management.
As PPR is a category A disease, pending confirmation, an early and conservative response is required which, under Regulation (EU) 2016/429 and Delegated Regulation 2020/687, includes movement bans, tracing, the establishment of protection and surveillance zones, and isolation of suspect infected animals (Box 2).
| Isolate the animal(s) showing suspected clinical signs |
| Halt all movements |
| Notify the veterinary authorities and activate the diagnostic system |
| Initiate tracing of animal movements |
Box 2. Actions required in case of suspicion
In the event of confirmation, the same regulation provides for stringent measures on the outbreak, aimed at eradicating the disease, as illustrated in Box 3.
| Seizure of the holding with immediate halt of all movements and initiation of the epidemiological investigation |
| Immediate notification of the event to the European Commission via the Animal Diseases Information System (ADIS), integrated with the WOAH international notification system (WAHIS)18 |
| Culling of all susceptible animals present in the epidemiological unit, followed by safe management and disposal of carcasses and contaminated materials, and cleaning and disinfection of all premises, equipment, and vehicles under official supervision |
| Establishment of protection zones (3 km radius) and surveillance zones (10 km radius) around the outbreak, within which intensified veterinary surveillance is implemented |
Box 3. Actions required in case of confirmation
Implications for Italy and Italy's response to the PPR risk
In this context of growing infectious pressure, Italy's disease-free status represents a condition to be preserved through informed and proactive actions. On one hand, the absence of notifications in the European WAHIS system confirms current freedom from disease; on the other, it highlights the country's vulnerability. The geographical position of the peninsula, only partially protected by natural barriers, exposes it to a multifactorial spillover risk. Proximity to the new Balkan outbreaks, particularly those in Croatia, constitutes a direct and immediate threat. Trans-Adriatic trade, both legal and illegal, represents a potential channel for silent introduction. Added to this is the significant density of small ruminant farms in some regions of north-eastern Europe, which, once the virus is introduced, facilitates its rapid and widespread dissemination. The combination of these factors – geographical proximity, intensity of trade, and susceptibility of the animal population – justifies the need to maintain a high level of attention, especially in terms of early clinical suspicion recognition, rapid activation of the diagnostic pathway, timely application of control measures, and communication management throughout the supply chain. This translates into maintaining a high level of alert among all stakeholders: from farmers to private veterinarians, up to the Experimental Zooprophylactic Institutes (IZSs).
For this reason, Italy has activated an extraordinary surveillance system aimed at preventing the introduction of the virus into the national territory. The Ministry of Health, through the Directorate General for Animal Health (DGSAF), issued an operational circular in July 2024 that raised the level of attention on all imports of small ruminants from epidemiologically affected areas, recommending targeted controls19. The system is structured on multiple levels, involving various components of the public veterinary network. In the first instance, the Veterinary Offices for EU Affairs (UVAC), peripheral structures of the Ministry of Health responsible for monitoring intra-Community trade, proceeded to analyse commercial flows recorded in the TRACES system (Trade Control and Expert System – the European Commission's online platform for mandatory sanitary and phytosanitary certification in trade of animals, animal products, food, feed, plants, and plant products), identifying consignments of small ruminants from countries with active outbreaks. For each shipment, the Veterinary Service of the territorially competent Local Health Authority (ASL) was activated, tasked with verifying the accompanying documentation and subjecting the animals to clinical examination, paying particular attention to symptoms compatible with PPR.
In the presence of suspected cases, the protocol provides for the involvement of the National Reference Centre for Exotic Animal Diseases (CESME), at the IZS of Abruzzo and Molise "G. Caporale," Teramo. CESME, as the National Reference Laboratory, constitutes the centre of scientific expertise for confirming any positive cases. In parallel, to strengthen field competencies, the Ministry and CESME have organized training initiatives in the form of webinars aimed at public veterinarians, with the objective of improving early disease recognition and the correct management of suspected cases.
The effectiveness of this framework lies in the synergy between the different components of the network, where the Ministry performs a coordination function, the UVAC and local Veterinary Services have a role in surveillance and intervention on the territory, while the IZSs and CESME carry out specialized diagnostic activities. This integrated system allows Italy to maintain constant risk monitoring, ensuring the ability to quickly trace at-risk consignments and implement, if necessary, targeted measures to protect the national livestock from a disease with a strong economic and health impact.
Conclusions
The developments of 2024–2025 have represented a decisive turning point in the history of Peste des petits ruminants in Europe, showing that the disease has become a concrete threat to previously disease-free contexts, imposing a paradigm shift in management. The central message is that, in a world hyperconnected by commercial flows and animal movements, disease-free status is not a permanent condition, but a goal to be preserved through constant investment. For Italy and for other European countries still free from the disease, the strategy must be based on an integrated model of proactive surveillance, which includes:
- Border controls and prevention
- Passive and active surveillance, to ensure early detection
- Efficient data flow and a rapid diagnostic network, capable of providing certainty in timely manner
- Standardized emergency plans, to ensure an immediate, coordinated, and effective response
- Intelligence provided by molecular epidemiology, to support control measures.
The ultimate goal of the global eradication strategy, targeted for 2030, remains valid, but, considering the events of the last two years, the path appears more complex. Preventing the introduction of the disease into still disease-free states and eradicating existing outbreaks today depends on the ability to transform knowledge of the disease and its epidemiology into effective and structured operational choices in terms of prevention and control.
The persistence of active outbreaks in the Balkan area and South-Eastern Europe confirms that the infectious pressure at the gates of the Union remains high. In this context, the risk of introduction through animal movement, even informal or illegal, makes it necessary to maintain a high level of alert through structured and widespread veterinary surveillance systems, especially for geographically exposed countries such as Italy.Acknowledgement
The authors would like to thank Dr Francesca Dall’Acqua for providing technical materials and for the critical review of the manuscript.
References
- FAO and OIE, 2015. Global Strategy for the Control and Eradication of PPR
- Ministero della Salute (2025) Piano Nazionale per le Emergenze di Tipo Epidemico: Manuale Operativo - Peste dei Piccoli Ruminanti. Versione Rev. 2.0, Ottobre 2025. Roma: Direzione Generale della Salute Animale, Ufficio 3 - Centro Nazionale di lotta ed emergenza contro le malattie animali. In collaborazione con: Centro di Referenza Nazionale per le Malattie Esotiche degli Animali (CESME), Istituto Zooprofilattico Sperimentale dell'Abruzzo e del Molise "G. Caporale"
- European Parliamentary Research Service. "Peste des petits ruminants: A race against time for livelihoods and livestock." July 9, 2025
- Unione Europea. (2009). Regolamento (CE) n. 1069/2009 del Parlamento Europeo e del Consiglio, del 21 ottobre 2009, recante norme sanitarie relative ai sottoprodotti di origine animale e ai prodotti derivati non destinati al consumo umano e che abroga il regolamento (CE) n. 1774/2002 (regolamento sui sottoprodotti di origine animale). Gazzetta ufficiale dell'Unione Europea, L 300, 1-33
- Unione Europea. (2016). Regolamento (UE) 2016/429 del Parlamento Europeo e del Consiglio, del 9 marzo 2016, relativo alle malattie animali trasmissibili e che modifica e abroga taluni atti in materia di sanità animale (legge sulla salute animale). Gazzetta ufficiale dell'Unione Europea, L 84, 1-208
- Commissione Europea. (2020). Regolamento delegato (UE) 2020/687 della Commissione, del 17 dicembre 2019, che integra il regolamento (UE) 2016/429 del Parlamento europeo e del Consiglio per quanto riguarda le norme relative alla prevenzione e al controllo di determinate malattie elencate. Gazzetta ufficiale dell'Unione Europea, L 174, 64-139
- Padhi, A. and Ma, L. (2014). Genetic and epidemiological insights into the emergence of peste des petits ruminants virus (PPRV) across Asia and Africa. Scientific Reports, 4, p. 7040. doi:10.1038/srep07040
- European Commission, Directorate-General for Health and Food Safety. (2024-2026). Presentations of the PAFF Committee on Animal Health and Welfare
- World Organisation for Animal Health (WOAH). (2024-2025). WAHIS event management: Peste des petits ruminants in Europe
- Commissione Europea. (2024). Decisione di esecuzione (UE) 2024/2923 della Commissione, del 20 novembre 2024, che modifica la decisione di esecuzione (UE) 2024/2132 relativa ad alcune misure di emergenza provvisorie contro l'infezione da virus della peste dei piccoli ruminanti in Grecia. Gazzetta ufficiale dell'Unione Europea, L 2923
- Kirtzalidou A, Bataille A, et al. (2025). Identification of peste des petits ruminants virus, Greece, 2024. Research in Veterinary Science, 194, 105840
- Commissione Europea. (2024). Decisione di esecuzione (UE) 2024/2154 della Commissione, del 6 agosto 2024, che modifica la decisione di esecuzione (UE) 2024/2119 relativa ad alcune misure di emergenza provvisorie contro l'infezione da virus della peste dei piccoli ruminanti in Romania. Gazzetta ufficiale dell'Unione Europea, L 2154
- Iacobescu-Marițescu I, et al. (2025). Field-based characterization of peste des petits ruminants virus outbreaks in Romania, 2024-2025. Transboundary and Emerging Diseases, 72(3), 1125-1138
- Commissione Europea. (2025). Decisione di esecuzione (UE) 2025/255 della Commissione, del 31 gennaio 2025, relativa ad alcune misure di emergenza provvisorie contro l'infezione da virus della peste dei piccoli ruminanti in Ungheria. Gazzetta ufficiale dell'Unione Europea, L 255
- SIVeMP - Società Italiana Veterinari per gli Animali da Reddito e da Compagnia. (2025). Focolaio Peste dei piccoli ruminanti in Ungheria. sivemp.it, 30 gennaio
- European Union Reference Laboratory for Peste des Petits Ruminants (EURL-PPR). (2025, August). Update on PPR emergence in Europe: Insights from viral genetic investigations [Conference presentation]. 1st GF-TADs Regional Conference in the European Region, Belgrade, Serbia. CIRAD
- Guendouz S, Bataille A, et al. (2025). Genomic analysis of peste des petits ruminants virus emergence in Greece, Romania and Bulgaria, 2024. Infection, Genetics and Evolution, 118, 105712
- European Parliamentary Research Service (EPRS). (2025). Peste des petits ruminants: The EU's response to outbreaks in South-East Europe (Briefing PE 775.865). European Parliament
- Ministero della Salute, Direzione Generale della Sanità Animale e dei Farmaci Veterinari (DGSAF). (2024). Notifica focolai di Peste dei piccoli ruminanti (PPR) in Grecia e Romania. Nota del Direttore Generale Giovanni Filippini, 24 luglio 2024.
Selene Corsini
Centro Operativo Veterinario per l’Epidemiologia, Programmazione, Informazione e Analisi del Rischio (COVEPI), Istituto Zooprofilattico Sperimentale dell’Abruzzo e del Molise “G. Caporale”, Teramo